This picture explains the idea of "atomic mass". The carbon atom (
14C) nucleus on the top has 6 protons plus 8 neutrons. It has an atomic mass of 14. Tritium (
3H), an isotope of hydrogen, is shown on the bottom. It has 1 proton plus 2 neutrons in its nucleus. Tritium has an atomic mass of 3.
Click on image for full size
Original artwork by Windows to the Universe staff (Randy Russell).
Atomic Number
Every atom has a nucleus. The nucleus has protons and neutrons in it. Scientists have a special name for the number of protons in an atom. They call it the "atomic number".
There are almost 100 different elements, like carbon and oxygen and gold. Each element has a different atomic number. Hydrogen atoms have 1 proton, so they have an atomic number of 1. Carbon has 6 protons and an atomic number of 6; oxygen has 8 protons and thus and atomic number of 8. The atomic number of uranium is 92!
Atoms of one kind of element always have the same number of protons. They often have different numbers of neutrons, though. Most carbon atoms have 6 protons and 6 neutrons. A few carbon atoms have 6 protons and 8 neutrons. The different kinds of atoms of the same element are called "isotopes".
Sometimes scientists talk about the "atomic mass" of an atom. If we add up the number of protons plus the number of neutrons we get the atomic mass. The atomic mass of the isotope of carbon that has 8 neutrons is 14 ( = 6 protons + 8 neutrons). Sometimes scientists use the letter "Z" to stand for atomic number and the letter "A" to stand for atomic mass.
You might also be interested in:

An element (also called a "chemical element") is a substance made up entirely of atoms having the same atomic number; that is, all of the atoms have the same number of protons. Hydrogen, helium, oxygen,
...more
Everything you see around you is made of tiny particles called atoms. Atoms are so small that you cant see them but scientists have found out that there are many different types of atoms, and each type
...more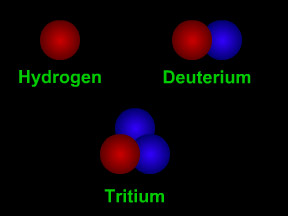
Isotopes are different "versions" of an element. All atoms of an element have the same number of protons. All hydrogen atoms have one proton, all carbon atoms have 6 protons, and all uranium atoms have
...more
One way scientists measure the size of something is by its mass. Mass is sort of like weight. Scientists can even measure very, very tiny things like atoms. One measure of the size of an atom is its "atomic
...more
Carbon-14 is an isotope of the element carbon. All carbon atoms have 6 protons in their nucleus. Most carbon atoms also have 6 neutrons, giving them an atomic mass of 12 ( = 6 protons + 6 neutrons). Carbon-14
...more
Looking for online content that can be used for a climate change education course or module? Pages linked below can be used to support an introductory climate change education for either a unit or a full
...more
Did you know that a lot of what happens in space physics has to do with the interaction of really tiny particles? These particles like protons and electrons are so tiny that you can't even see them with
...more












