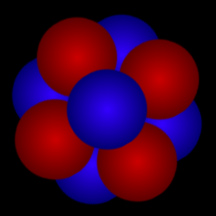
Although the nucleus of an atom is far too small for us to see, here's one way of thinking about an atomic nucleus: as a cluster of tightly packed "balls". The red "balls" represent protons; the blue "balls" represent neutrons. The cloud of electrons that "orbit" an atom's nucleus and define the "size" of an atom is roughly 100,000 times as large as that atom's nucleus!
Click on image for full size
Original artwork by Windows to the Universe staff (Randy Russell).
Atomic Nucleus
Atoms have two main parts: a massive nucleus at the center and a swarm of fast-moving electrons around it. The nucleus is made up of protons and (usually) neutrons. Almost all of the mass (more than 99%) of an atom is in the dense nucleus.
An atomic nucleus is much, much smaller than an atom. The cloud of electrons that "orbit" the nucleus and define the "size" of an atom is about 100,000 times as large as that atom's nucleus! If you made a scale model of an atom with a nucleus the size of a pea, the electrons would zing around in a space larger than a major sports stadium! An atom is mostly empty space.
You might also be interested in:

One way scientists measure the size of something is by its mass. Mass is sort of like weight. Scientists can even measure very, very tiny things like atoms. One measure of the size of an atom is its "atomic
...more
Every atom has a nucleus. The nucleus has protons and neutrons in it. Scientists have a special name for the number of protons in an atom. They call it the "atomic number". There are almost 100 different
...more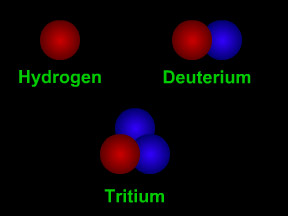
Isotopes are different "versions" of an element. All atoms of an element have the same number of protons. All hydrogen atoms have one proton, all carbon atoms have 6 protons, and all uranium atoms have
...more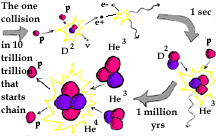
All the gory details ..... First two protons collide and form a deuterium nucleus (one proton + one neutron). This is not as simple as it sounds. Only one collision in ten trillion trillion actually produces
...more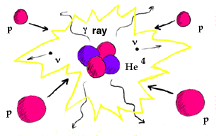
The sun uses hydrogen as fuel mostly through a series of nuclear reactions called the proton-proton chain, leaving behind helium ash. Just the bare facts .... In the proton-proton chain, 4 protons are
...more
"But wait a minute," you say. "We've tried this nuclear fusion stuff on Earth to produce energy and so far it hasn't worked very well. How does the sun succeed where we have failed?" You are right. Operational
...more
There are four forces in the Universe: strong, weak, electromagnetic and gravity. You may be most familiar with gravity. Gravity is what holds you and your dog to the surface of this planet! The other
...more