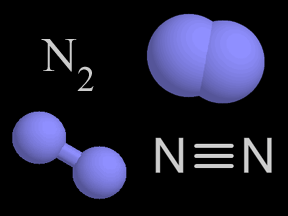This drawing represents a comet bringing atmospheric molecules and possibly primitive life forms to the Earth's surface.
Click on image for full size
JPL/NASA
Base
Like an
acid, a
base is a substance capable of dissolving things. Unlike an acid in which the active agent is a substance which has a positive electric charge (H
+), in a base the active agent has a negative charge (OH
-). Here are a couple of scientific explanations of what a base is:
A base is any substance that upon dissolving in water increases the hydroxide ion content (OH-). An base can also be thought of as a substance which accepts a proton from another species in a reaction. (Here, "accept" is a passive term for what really happens. A base attacks and "rips" a proton from a substance which has any capability of giving it away. Thus a base accepts a proton the way a thief "accepts" your purse). Bases are classified as either strong or weak depending upon their relative ability to do this.
There are many bases. Here are a few: Ammonia (used as a cleaner), Calcium hydroxide (used in mortar for construction), Magnesium hydroxide (otherwise known as milk of magnesia; used as a laxative or antacid), Sodium hydroxide (used as an oven cleaner).
You might also be interested in:
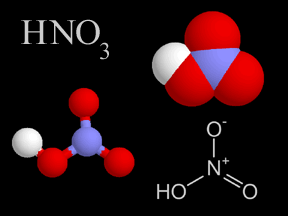
Nitric acid is a colorless, corrosive liquid and a toxic acid which can cause severe burns. Nitric acid consists of nitrogen, oxygen, and hydrogen atoms. Nitric acid, in its gas phase, is present in very
...more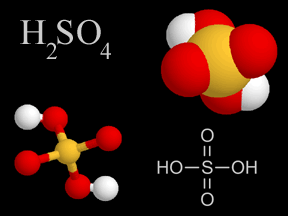
Sulfuric acid is a viscous, oily liquid and a strong acid which can cause severe burns. Sulfuric acid consists of sulfur, oxygen, and hydrogen atoms. Sulfuric acid is one of the components of acid rain.
...more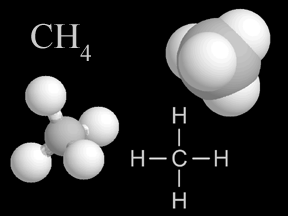
Methane is gas that is found in small quantities in Earth's atmosphere. Methane is the simplest hydrocarbon, consisting of one carbon atom and four hydrogen atoms. Methane is a powerful greenhouse gas.
...more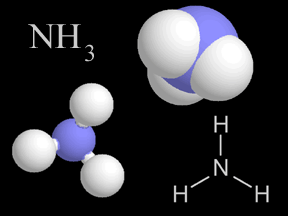
Ammonia is a chemical compound that is a colorless, flammable gas at normal temperatures and pressures. Ammonia is toxic, corrosive to some materials, and has a pungent odor. An ammonia molecule (NH3)
...more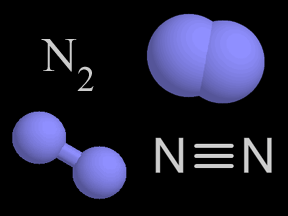
Nitrogen is a chemical element with an atomic number of 7 (it has seven protons in its nucleus). Molecular nitrogen (N2) is a very common chemical compound in which two nitrogen atoms are tightly bound
...more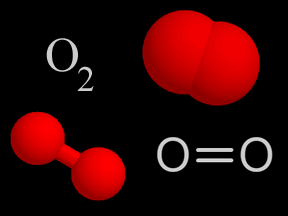
Oxygen is a chemical element with an atomic number of 8 (it has eight protons in its nucleus). Oxygen forms a chemical compound (O2) of two atoms which is a colorless gas at normal temperatures and pressures.
...more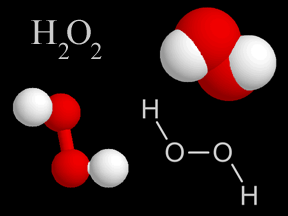
Hydrogen peroxide (H2O2) is a chemical compound consisting of two oxygen atoms and two hydrogen atoms. It is a clear liquid at normal temperatures and pressures. Hydrogen peroxide is a very reactive chemical.
...more