This is a picture of the meteorite ALH84001.
Click on image for full size
NASA
The possible discovery of Life on Mars
In July, 1996, it was announced that Dr. David McKay, along with a team of
scientists at Johnson Space Center (a division of NASA), had discovered
possible fossils of
bacteria in a meteorite named ALH84001 that came from Mars. It was found in the Allen Hills in Antarctica in 1984 after having landed there 12,000 years ago. While many scientists were excited at first, much of the proof offered fell apart. NASA said that after two years of study "a number of lines of evidence have gone away".
Several different chemicals and molecular structures were exciting because they looked similar to byproducts of life on Earth. However, these chemicals and structures can also be created without life. Some are even present in deep space on comets, and scientists do not think that they came from Martian life anymore.
Small spheres were observed in the meteorite which the
scientists in 1996 claimed were the fossilized remains of bacteria. However, they are roughly 1000 times smaller than the smallest bacteria on Earth, so don't resemble any life thought to be possible. Organic (carbon containing) compounds were found with the spheres, but it turned out that the organic compounds became a part of the meteorite after it landed on Earth (possibly when water seeped in a couple times over the 12,000 years the rock laid in Antarctica). Carbon 14, an isotope found on Earth is present in the organic compounds, but not in the spheres.
The environment of Mars in the
past was very different than it is today. Conditions then may have been favorable for the existence of life. Even though the Mars meteorite does not prove life once existed on Mars, it certainly does not disprove the possibility.
You might also be interested in:
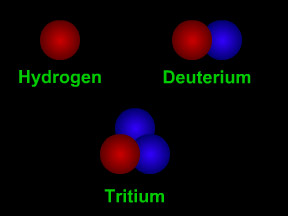
Isotopes are different "versions" of an element. All atoms of an element have the same number of protons. For example, all hydrogen atoms have one proton, all carbon atoms have 6 protons, and all uranium
...more
Jupiter's atmospheric environment is one of strong gravity, high pressure, strong winds, from 225 miles per hour to 1000 miles per hour, and cold temperatures of -270 degrees to +32 degrees (freezing temperature).
...more
In July, 1996, it was announced that Dr. David McKay, along with a team of scientists at Johnson Space Center (a division of NASA), had discovered possible fossils of bacteria in a meteorite named ALH84
...more
Saturn's atmospheric environment is one of strong gravity, high pressure, strong winds, from 225 miles per hour to 1000 miles per hour, and cold temperatures of -270 degrees to +80 degrees. With winds
...more
Titan's atmosphere is a lot like the Earth's, except that it is very cold, from -330 degrees to -290 degrees! Like the Earth, there is a lot of Nitrogen and other complex molecules. There also may be an
...more
Autotrophs are organisms that can "make their own food" from an inorganic source of carbon (carbon dioxide) given a source of energy. Most autotrophs use sunlight in the process of photosynthesis to make
...more
In the warm primordial ocean, aggregates of amino acids, proteins, and other hydrocarbons came together into a form called *coacervates*. Amino acids will spontaneously form coacervates in the same way
...more














