Click on image for full size
Original artwork by Windows to the Universe staff (Randy Russell).
Related links:
Kelvin Temperature Scale
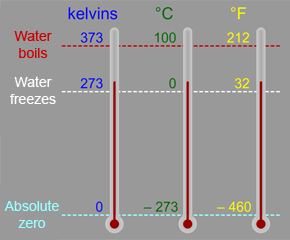
The Kelvin scale is a temperature scale that is often used in astronomy and space science. You are probably more familiar with the Celsius (or Centigrade) scale, which is part of the metric system of measures, and the Fahrenheit scale, which is used in the English system.
The Kelvin scale is similar to the Celsius scale. Zero degrees is defined as the freezing point of water in the Celsius system. However, the zero point in the Kelvin scale is defined as the coldest possible temperature, known as "absolute zero". Absolute zero is –273.15° C or –459.67° F. The "size" of a one degree change in temperature is exactly the same in the Celsius and Kelvin scales, so the freezing point of water is at a temperature of 273.15 kelvins (that is, 273.15 degrees above absolute zero). Water boils at a temperature of 100° C, which is 373.15 kelvins (or 212° F).
Notice how we don't say "degrees Kelvin" like we would say "degrees Celsius" or "degrees Fahrenheit". Instead of saying a temperature is "300 degrees Kelvin", we say the temperature is "300 kelvins".
To convert a temperature measurement between the Kelvin scale and the Celsius or Fahrenheit scales, use the following formulas:
| K = C + 273.15 | or | C = K - 273.15 | ||
| K = (F + 459.67) / 1.8 | or | F = 1.8 K - 459.67 |
... where K stands for the temperature in kelvins, C for temperature in degrees Celsius, and F for temperature in degrees Fahrenheit.
The Kelvin scale was named after William Thomson, Lord Kelvin, who was a prominent Irish/Scottish physicist and engineer of the latter part of the 19th century. Thomson did important work on the physics of heat as well as other disciplines within the physical sciences.