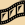Photodissociation of the Hydroxyl Radical (OH)
|
The hydroxyl radical (OH) is sort of a piece of a molecule. It has one oxygen atom and one hydrogen atom. One way to make OH is to knock a hydrogen atom off of a water molecule (H2O).
This movie shows how ultraviolet "light" can break apart the hydroxyl radical. You need the latest version of the Flash player to see this movie.
Photons are little bits of light. Photons carry energy.
When a photon hits the hydroxyl radical, it adds energy to the radical. The radical has chemical bonds (they work a bit like stretchy rubber bands!) holding its atoms together. Sometimes the energy from the photon breaks the bond between the oxygen atom and the hydrogen atom. The atoms fly apart!
When a photon knocks molecules (or the hydroxyl radical) apart, it is called photodissociation.
Photons of ultraviolet (UV) light carry more energy than photons of visible light carry. It takes high-energy UV photons to break the hydroxyl radical apart!
|
 Photodissociation of Water
Photodissociation of Water
 Photodissociation of Oxygen
Photodissociation of Oxygen
 Photodissociation of Nitrogen
Photodissociation of Nitrogen
You might also be interested in:
Most things around us are made of groups of atoms connected together into packages called molecules. Molecules are made from atoms of one or more elements. Some molecules are made of only one type of
...more
Text for this level has not been written yet. Please see the "Intermediate" text for this page if you want to learn about this topic. To get to the "Intermediate" text, click on the blue "Intermediate"
...more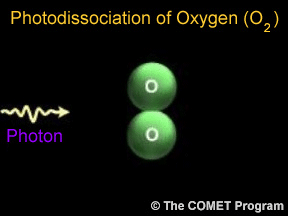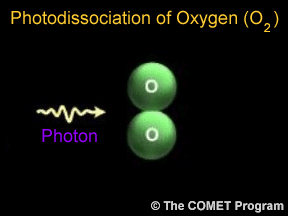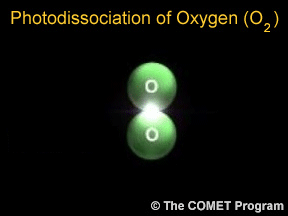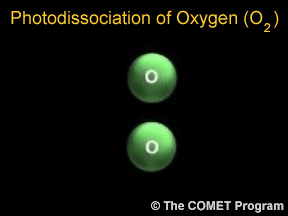
Photons are little bits of light. Photons carry energy. When a photon hits a molecule, it adds energy to the molecule. The molecule has chemical bonds (they work a bit like stretchy rubber bands!) holding
...more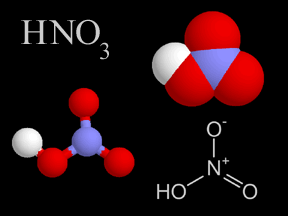
Nitric acid is a very strong kind of acid. If you got some on your skin, it would burn you! Nitric acid has nitrogen, oxygen, and hydrogen atoms in it. Earth's atmosphere has a very, very tiny bit of nitric
...more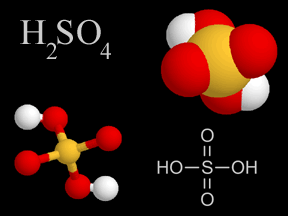
Sulfuric acid is a very common type of acid. Acid rain has sulfuric acid in it. Acid rain harms plants, fish, and other living things. A type of air pollution causes acid rain. When people burn fossil
...more
Methane is a kind of gas. There is a small amount of methane in the air you breathe. A methane molecule has carbon and hydrogen atoms in it. Methane is a greenhouse gas. That means it helps make Earth
...more
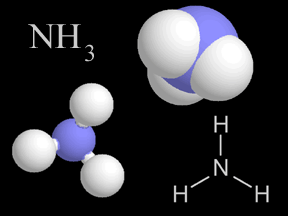
Ammonia is a kind of gas. Ammonia molecules (NH3) have hydrogen and nitrogen atoms in them. The air you breathe has a tiny bit of ammonia in it. When plants and animals die and decay, they give off ammonia.
...more