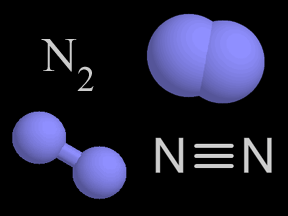This picture shows radioactive decay of a carbon-14 atom. The carbon atom gives off a beta particle of radiation. The carbon atom turns into a nitrogen atom.
Click on image for full size
Original artwork by Windows to the Universe staff (Randy Russell).
Carbon-14 Dating
Carbon-14 dating (also called "radiocarbon dating") is used to determine the age of materials that contain carbon that was originally in living things. It is often used in archeology and some types of biology. Living creatures ingest carbon. Plants (and other autotrophs) take in carbon dioxide gas from the atmosphere during photosynthesis. Animals (and other heterotrophs) get their carbon by eating plants or other animals, from decaying organic matter, or from other similar sources.
Some of the carbon is a radioactive isotope called carbon-14 (14C). When the creature dies, it stops ingesting carbon. The radioactive 14C gradually undergoes radioactive decay, transforming it into nitrogen, and therefore gradually "disappears". Scientists can study samples from the once-live creatures' remains to see how much radioactive 14C, as compared to the normal isotope of carbon (carbon-12 or 12C), is still around. This tells the scientists how long ago the organism died.
14C has a half-life of 5,730 years, meaning that after about five thousand years about half of the 14C will decay and turn into nitrogen. After several half-lives, too little 14C will remain in a sample for it to be useful for dating. Radiocarbon dating is therefore only useful for samples with ages of less than about 65,000 to 80,000 years.
You might also be interested in:

Autotrophs are organisms that can "make their own food" from an inorganic source of carbon (carbon dioxide) given a source of energy. Most autotrophs use sunlight in the process of photosynthesis to make
...more
Photosynthesis is the name of the process by which autotrophs (self-feeders) convert water, carbon dioxide, and solar energy into sugars and oxygen. It is a complex chemical process by which plants and
...more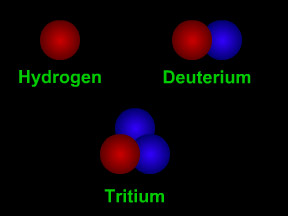
Isotopes are different "versions" of an element. All atoms of an element have the same number of protons. For example, all hydrogen atoms have one proton, all carbon atoms have 6 protons, and all uranium
...more
Carbon-14 is an isotope of the element carbon. All carbon atoms have 6 protons in their nucleus. Most carbon atoms also have 6 neutrons, giving them an atomic mass of 12 ( = 6 protons + 6 neutrons). Carbon-14
...more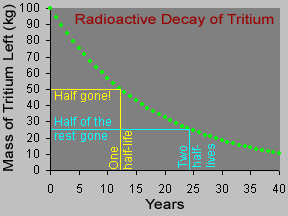
Some materials are radioactive. Their atoms give off radiation. When an atom gives off radiation, it turns into a different kind of atom. That is called radioactive decay. Some atoms decay very quickly,
...more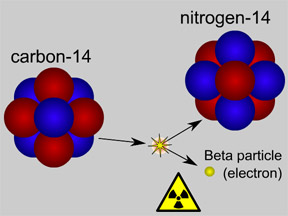
Some materials are radioactive. They emit radiation. When an atom of a radioactive substance gives off radiation, it becomes a new type of atom. This process is called radioactive decay. There are two
...more
There is more nitrogen gas in the air than any other kind of gas. About 4/5ths of Earth's atmosphere is nitrogen gas! A molecule of nitrogen gas is made up of two nitrogen atoms. There are other molecules
...more