Click on image for full size
L.Gardiner/Windows to the Universe
Chemistry
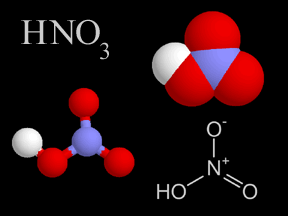
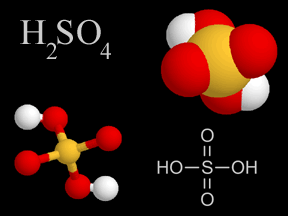
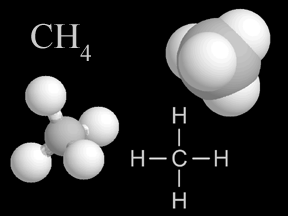
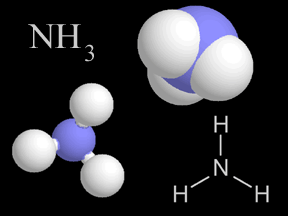
Chemistry is the study of matter, energy, and their interactions. Chemists study the composition of substances, their properties, and how they react with each other under varying circumstances. Indeed, chemists work to develop new substances, with properties that help us solve problems faced by society today.Because chemistry deals with the fundamental structure of matter (such as the structure of the atom, electrons, protons, and neutrons), it overlaps with physics. Because chemistry is involved in the biology of life (for instance, in photosynthesis), it overlaps with biology. Similarly, because geology involves study of the matter the Earth and other bodies in the solar system are made of, geology involves chemistry.
Geochemistry is the study the properties of Earth materials, working to understand how they vary under different pressures and temperatures. Geochemists study the abundances of the chemical elements in minerals, soils, rocks, and ores, as well as in the Earth's hydrosphere and atmosphere. They study how these elements move and change due to processes in the Earth system.
Biogeochemistry is the study of the chemical composition of substances in the natural environment, and how they change based on chemical, physical, geological, and biological processes in the Earth system. Biogeochemists study cycles in the Earth system, such as the carbon cycle and nitrogen cycle, to understand the role of these substances in the Earth system.
The origin of the word "chemistry" is not clear. It appears to have evolved from the word "alchemy", which may come from the Arabic word "al-kimia". Alchemy was the practice of trying to change common metals into gold, and attempts to develop other magical compounds that would allow immortality or ultimate wisdom.