A student from the HIGH TIDE project turns on the CTD instrument. High school students use the CTD recorder to measure salinity, temperature and depth of the water in the Lafayette River which is a part of Chesapeake Bay.
Click on image for full size
Image courtesy of the HIGH TIDE project
Salinity - Dissolved Salts, Measuring Salinity
When we measure the salinity of water, we look at how much dissolved salt is in the
water, or the concentration of salt in the water. Concentration is the amount (by weight) of salt in water and can be expressed in parts per million (ppm). Here are the classes of water:
- Fresh water - less than 1,000 ppm
- Slightly saline water - From 1,000 ppm to 3,000 ppm
- Moderately saline water - From 3,000 ppm to 10,000 ppm
- Highly saline water - From 10,000 ppm to 35,000 ppm
Ocean water has a salinity that is approximately 35,000 ppm. That's the same as saying ocean water is about 3.5% salt. Sometimes, salinity is measured in different units. Another common unit is the psu (practical salinity units). Ocean water has a salinity of approximately 35 psu. Scientists measure salinity using a CTD instrument (CTD = conductivity, temperature, depth).
Ocean water is about 3.5% salt. That means that if the oceans dried up completely, enough salt would be left behind to build a 180-mile-tall, one- mile-thick wall around the equator. About 90 percent of that salt would be sodium chloride, or ordinary table salt. Chlorine, sodium and the other major dissolved salts of the ocean are listed in this table:
Dissolved salts in
sea water (atoms): |
| 55.3 % Chlorine |
| 30.8 % Sodium |
| 3.7 % Magnesium |
| 2.6 % Sulfur |
| 1.2 % Calcium |
| 1.1 % Potassium |
You might also be interested in:
About 70% of the Earth is covered with water. Over 97% of that water is found in the oceans. Everyone who has taken in a mouthful of ocean water while swimming knows that the ocean is really salty! Dissolved
...more
The Phoenix Mars Lander was a robot spacecraft that was sent to Mars. Phoenix landed near the North Pole on Mars. This page tells about the mission of Phoenix. It also describes the instruments on the
...more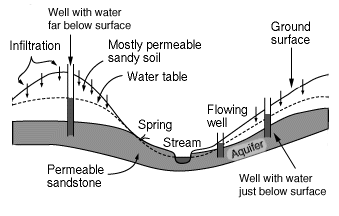
An aquifer is the name for a layer of rock which is capable of holding a large amount of water. Some layers are better at holding water than others, for example a layer of sandstone can hold a good deal
...more
Carbonate is a name for rocks and minerals which contain a molecule made of both carbon and oxygen known as CO32-. (CO32- is also known as the molecule carbonate). Limestone is an example of a calcium
...more
One process which transfers water from the ground back to the atmosphere is evaporation. Evaporation is when water passes from a liquid phase to a gas phase. Rates of evaporation of water depend on things
...more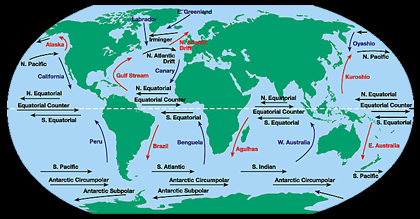
The water at the ocean surface is moved primarily by winds that blow in certain patterns because of the Earths spin and the Coriolis Effect. Winds are able to move the top 400 meters of the ocean creating
...more
Rivers are very important to Earth because they are major forces that shape the landscape. Also, they provide transportation and water for drinking, washing and farming. Rivers can flow on land or underground
...more















